Final Results and Investor Presentation
Latest Results
Audited Final Results for the period ended 31 December 2025
ProBiotix Health plc (AQSE: PBX), the life sciences business developing probiotics to support cardiometabolic health, announces its audited results for the period ended 31 December 2025.
|
Download The full results are available
in |
Key Highlights
- Sales +45% to £2.73m (2024: £1.88m)
- Gross Profit +46% to £1.48m (2024: £997,000)
- EBITDA loss from continuing operations down 34% to £426,000* (2024: £642,000*)
- Gross Profit margin improved to 54% (2024: 53%)
- Group remains fully funded with a cash balance of £1.27m
- 10 new products launched during the year
*Before share based payment expenses
Post period-end
- During Q1 2026 the Group has achieved profitability
- Outlook positive with a record Q1 2026 order book +110% at £1.3m
- Strong momentum in China, with increasing sales channels being delivered by partner Kemin China Technology
- Recent launch of new women’s health products is gaining further tangible traction - well placed to target an accessible B2B market worth more than $5 billion
- Planned (and funded) expansion in new metabolic health areas to include prediabetes studies
- Further margin improvement has been achieved, driven by CMO optimisation
Investor Presentation
The Company is hosting a presentation for investors via the Investor Meet Company platform today at 10:00 am BST. The presentation is open to all existing and potential shareholders. Questions can be submitted pre-event via your Investor Meet Company dashboard, up until 9:00 am BST today, or at any time during the live presentation.
To sign up to the presentation via Investor Meet Company please register using the following link: https://www.investormeetcompany.com/probiotix-health-plc/register-investor
Steen Andersen, CEO of ProBiotix Health, commented: “The current year has started positively, with a record Q1 order book of £1.3 million, reflecting a broader customer base, new contract wins and increased reorder activity.
The Group intends to prioritise further expansion across the Asia-Pacific region, with the Kemin partnership in China expected to contribute increasingly to revenue growth, while also providing geographical diversification.
In Europe, potential regulatory developments relating to cholesterol-lowering ingredients are creating a number of opportunities for LPLDL®. and in the United States, we expect a number of late-stage projects to convert into commercial agreements.
The achievement of Q1 profitability is a significant step change for the business and is testimony to the hard work of the team. With a highly scalable business, record new enquiries, and a significant addressable market, we view the future with considerable confidence.”
Chairman and Chief Executive's Statement
FY 2025 represented a further year of progress in strengthening the Group’s position as a global innovator and commercial partner in probiotic-based cardiometabolic health supplements. The Company continued to execute against its stated strategy, delivering strong revenue growth while advancing operational scalability, and progressing towards profitability.
Revenue growth was achieved through a combination of geographic expansion, new customer acquisition and continued growth from existing customer relationships. During the year, the Group further expanded its presence in the United States and successfully entered the Chinese market, while continuing to invest selectively in infrastructure to support future scale.
The volume of new commercial projects initiated during early FY 2025 provided additional confidence in the relevance of the Group’s proprietary technology and its alignment with long-term consumer trends in cardiometabolic health and healthy aging.
Based on the Group’s performance and outlook, the Board and Management continue to believe that the Company’s underlying fundamentals are not yet fully reflected in its current market capitalisation. Comparisons with peer companies in the probiotics and health supplements sector indicate potential for a re-rating as the business continues to scale.
Results
The Group delivered meaningful progress towards profitability in FY 2025. Revenue increased by 45% to £2.73 million (2024: £1.88 million), reflecting continued commercial traction across core markets. Gross profit increased by 49% to £1.48 million (2024: £0.99 million), with gross margin improving to 54% (2024: 53%).
Other administrative costs increased modestly by 9% to £1.9 million, in line with management expectations and reflecting controlled investment in growth, together with inflationary pressures.
EBITDA loss from continuing operations, before share-based payment charges, reduced by 34% to £426,000 (2024: £642,000), demonstrating the operating leverage within the business model. The company has recognised a share based payments charge in the year of £753,000.
Having achieved the first milestone of £2.5m revenue in a 12 month period, the company are confident the remaining milestones will be achieved so the charge can be recognised over the period in which the targets will be achieved. The charge, which is not a cash cost to the company, is heavily weighted in the first year, the charge for the future years will be significantly reduced. The net loss for the year, including amortisation and share-based payment charges, was £1.2m (2024: £0.8m).
The Group ended the year with cash balances of £1.27 million (2024: £1.65 million), providing adequate resources to support our planned commercial and operational initiatives.
Strategic focus and Market Opportunity
ProBiotix is a life sciences company with a strategic focus on probiotics and the human microbiome addressing various aspects of cardiometabolic health, and other lifestyle conditions impacting the quality of life throughout the lifespan. The Company operates in the dietary supplement prevention segment, tapping into the mega consumer trend of healthy ageing, cardiometabolic health and probiotics. Our purpose is to provide consumers and industry worldwide with uncompromised supply and service of safe and scientifically validated probiotic-based microbiome product solutions for the improvement of human health. The Company aims to partner with scientifically driven dietary supplement suppliers, consumer health and Over The Counter (OTC) companies globally who have a defined strategic focus on the human microbiome. Our commercial focus is to provide private label, turnkey probiotic dietary supplements to target preventative cardiometabolic health and healthy ageing, taking off-set in the Company’s principal proprietary and patent protected probiotic strain LPLDL®. In selected markets we complement the value proposition further by including ingredient sales of LPLDL® as a means of accelerating short-term market penetration and building brand awareness.
Cardiovascular disease (CVD) remains the leading cause of deaths globally, ahead of cancer. The latest statistics from the World Heart Federation show that, CVD accounts for approximately 20.5 million deaths annually, representing around 33% of all deaths (Source: World Heart Federation | Global Heart Health & CVD Advocacy). There are an increasing number of women suffering from CVD, with up to 77% presenting elevated cholesterol levels and up to 5 times higher risk of heart attack during and after menopause (Source: Health Survey for England, 2021 part 2 - Adults’ health: Cholesterol). CVD has been identified as one of the key leading causes mortality in middle aged women (Sources: World Heart Federation |Global Heart Health & CVD Advocacy and American Heart Association; Circulation; Women’s Health Initiative).
Some 30% of all deaths in women are caused by CVD with the global number having risen by nearly 50% to almost 9 million deaths annually over the past 20 years (World Heart Federation | Global Heart Health & CVD Advocacy + Medicine. 104(27):e43215, July 04, 2025). According to the latest research from the World Heart Federation and American Heart Association ( Source: World Heart Federation | Global Heart Health & CVD Advocacy) between 3.6 to 4.4 million people worldwide succumb to high cholesterol annually, and approximately 39% of the global adult population are categorised as having elevated cholesterol, and thereby in the risk group of CVD. With 80% of all CVD deaths being preventable according to WHO and average life expectancy having increased by 6.2 years over the past 30 years (Institute for Health Metrics and Evaluation 2023) the category offers a unique positioning opportunity for ProBiotix Health’s product portfolio and technology in a fast expanding market for preventive supplements.
This underlying disease burden is increasingly translating into measurable consumer demand for preventive, science-backed health solutions. According to Innova 73% of global consumers consider healthy ageing “extremely” or “very important” in their day-to-day choices, reflecting a shift towards long-term health maintenance. (Source: Innova Insights)
In the UK, 45% of consumers agree that adhering to a health plan can help prevent future conditions (Source: Innova Market Insights) Source: Mckinsey & Company) Source: Euro Monitor) of global consumers identify naturalness as their most important eating practice, underscoring growing demand for clinically validated, naturally derived solutions.
The global probiotics supplement consumer market is, in its own right, forecast to reach $12.7 billion by 2028 (https://ipa-biotics.org/human-market.). Market growth is increasingly driven by consumer demand for high quality and clinically backed “precision probiotics” in the area of Cardiometabolic Health, Women’s Health and Healthy Ageing. The reduction of cardiometabolic disease-related risk factors remain one of the top priorities for legislators, and thus offers distinct growth opportunities for the category.
In addition, the global consumer market for preventive cardiometabolic health supplements is estimated to reach over $16 billion towards 2028 (Source: Precedence Research)
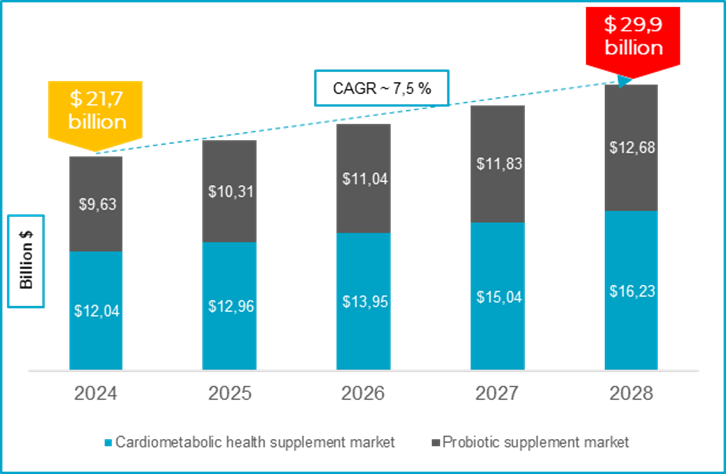
Source:
- https://www.precedenceresearch.com/cardiovascular-health-supplements-market
- https://ipa-biotics.org/human-market/
The top five market growth factors for cardiometabolic health supplements can be outlined as follows (source: Precedence Research):
📊 Rising Prevalence of Cardiovascular Disease (CVD)
👴 Aging Population
🛡️ Preventive Healthcare Focus
🌍 Growing Disposable Income in Emerging Markets
👩⚕️ Increased Consumer Awareness & Health Consciousness
As a means of targeting this lucrative market, leading brands are looking to add new products to their lineup based on novel innovative ingredients, and product solutions allowing for clear positioning and differentiation. As a provider of private label turnkey supplements, ProBiotix participates in approximately 20-25% of the value creation in the current $25 billion consumer market, representing a $5-$7.5 billion market potential. With a projected accumulated consumer market value of $30 billion by 2028, the cardiometabolic health and probiotic supplement markets continue to represent a significant opportunity for ProBiotix in a space with a limited number of scientific backed product solutions.
Key Milestone Achievements in FY 2025
- FY2025 delivered a satisfactory result against our stated strategy and operational objectives.
- The strong trading performance reflected robust commercial traction across our core markets, supported by both new customer acquisition, and continued positive growth from already established customer partnerships.
- We implemented a number of internal initiatives to enhance operational scalability, and enhance the path to profitability.
- Our recently established partnership with global ingredients specialist Kemin performed strongly during the year, benefiting from the execution of Kemin's highly effective market positioning strategy. The material outcome was realised at the end of the year, less than 12 months after market introduction, with their promising sales pipeline converting to commercial sales.
- The relationship with key customers remained robust, delivering continued growth while at the same time, we managed to further reduce the Company's dependency on a few key customers by the addition of potential cornerstone partners such as Wellizen (Australia), RevivaBio (Sweden) and NutriWow (China).


- Material progress towards profitability was reached during the year. EBITDA losses were reduced by 34% to £426,000 in 2025 (2024: £642,000), fully in line with our stated strategic objectives. The gross profit margin edged higher to 54% (2024: 53%), with multiple initiatives underway to further enhance margins in 2026 and beyond, including the conclusion of our regional Contract Manufacturing Organisation (CMO) strategy to improve supply security, closeness to market and cost optimisation.
- A new menopause-focused women's health concept, based on our LPLDL® strain - YourBiotixMH was launched. Early commercial traction has been positive, with 27 projects currently open, a number of which are in detailed discussion, and the first two product launches are anticipated for early 2026. The interest in these new products is driven by growing consumer focus on science-backed solutions in women's health and healthy ageing.
- Commercial traction continued to build throughout the year. In 2025 we generated a significant increase in enquiries, with over 280 'qualified leads' (2024: 190). This has been driven by our participation in major global industry events, including SupplySide Global (USA), Vitafoods Europe (Spain) and Vitafoods Asia (Thailand). The active customer portfolio expanded by approximately 50%, following the successful onboarding of new customers across multiple geographies.
- We also continued to strengthen our scientific and clinical foundations. Applications were submitted for ethical committee approval for two new clinical trials. The first, a three-year study expected to include over 200 participants, will investigate the potential benefits of LPLDL® in individuals with pre-diabetes, type I diabetes and type II diabetes. A successful outcome could support future expansion of LPLDL® into the preventive pre-diabetes category. The second trial, with approximately 60 participants, is designed to further reinforce the clinical evidence supporting the role of LPLDL® cardiometabolic health and cholesterol reduction.
- Geographic expansion progressed well during the year, with entry into the Korean market through the launch of our CholBiome X3, a finished product containing the Company's proprietary LPLDL® ingredient. The launch was executed in partnership with P.Bros, Korea's leading influencer-driven online pharmacy platform.
- The current five-year strategy plan was successfully updated and replaced with a new three-year strategy looking towards 2028.
Board and Management
There were no changes to the composition of the Board or senior management during FY 2025. The Board considers the current structure to be appropriate to support the Group’s near and medium-term objectives. Incremental additions may be made as the business continues to scale.
Outlook
FY 2026 commenced positively, with a record Q1 order book of £1.3 million, reflecting a broader customer base, new contract wins and increased reorder activity.
The Group intends to prioritise further expansion across the Asia-Pacific region, with the Kemin partnership in China expected to contribute increasingly to revenue growth, while also providing geographic diversification.
In Europe, potential regulatory developments relating to cholesterol-lowering ingredients are creating a number of opportunities for LPLDL®, The Company is engaged in several active replacement projects, with an initial major product launch expected in H1 2026
. In the United States, management expects a number of late-stage projects to convert into commercial agreements. While short-term macroeconomic and trade-related uncertainties may impact the pace of expansion, no material long-term impact is anticipated, supported by ongoing localisation of manufacturing.
The Group remains adequately funded to execute its growth strategy. Admission to the Apex Segment of the Aquis Stock Exchange during the year marked an important milestone, reflecting the Company’s scale and governance maturity, and supporting continued engagement with institutional and private investors.
With a highly scalable business, record new enquiries, and a significant market to address, we view the future with considerable confidence.
| A Reynolds | S Andersen |
| Chairman | Chief Executive Officer |
Date: 27 March 2026
REVIEW OF BUSINESS
A review of the business of the Group, together with comments on future developments is given in the Chairman’s and Chief Executive’s Statements.
PRINCIPAL RISKS AND UNCERTAINTIES FACING THE GROUP
Technology and products
The Group is involved in the discovery and development of microbiome modulation products. The development and commercialisation of its intellectual property and future products will require human nutritional studies and there is a risk that products may not perform as expected. This risk is common to all new products developed for human consumption.
Technologies used within the food, beverage and healthcare marketplace are constantly evolving and improving.
There is a risk that the Group’s products may become outdated or their commercial
value decrease as improvements in technology are made and competitors launch competing products. To mitigate this risk the Group is working with industry key opinion leaders, attends international conferences and has developed a research and development department which will keep up with the latest developments in the industry.
Intellectual Property
The Group is focused on protecting its IP and seeking to avoid infringing on third parties’ IP. To protect its products, the Group is building and securing patents to protect its key products. However, there remains the risk that the Group may face opposition from third parties to patents that it seeks to have granted and that the outstanding patent applications are not granted. The Group engages legal advisers to mitigate the risk of patent infringement and to assist with the protection of the Group’s IP.
KEY PERFORMANCE INDICATORS
Financial
|
|
Year to 31 December 2025 | Year to 31 December 2024 | |
| £’000 | £’000 | ||
| Revenue | 2,732 | 1,883 | |
| Operating Loss | (1,235) | (852) | |
| Loss for the period | (1,233) | (847) | |
| Cash as at 31 December 2025 | 1,266 | 1,646 |
During the year to 31 December 2025 the company has achieved a number of key objectives to build shareholder value, these are laid out in the Chairman and Chief Executive’s statement.
Non-financial
The Board recognises the importance of KPI’s in driving appropriate behaviour and enabling of Group performance. For the year to 31 December 2025 the primary KPI’s were to develop customer relationships established in 2024 to successful contract relationships and onboarding in FY 2025. In 2025 the customer portfolio expanded to 23 customers from 13 in 2024.
DIVIDENDS
No dividends are distributed for the year to 31 December 2025. (2024: NIL)
FUTURE DEVELOPMENTS
The Chairman’s and Chief Executive statement on gives information on the outlook of the Group.
Corporate Governance
Executive Management:
The Group’s current executive team comprises:
| A Reynolds | Non-Executive Chairman |
| S Andersen | Executive Director and CEO |
| S O’Hara | Non-Executive Director |
| M Caspani | Non-Executive Director |
| F Bruhn-Petersen | Non-Executive Director |
Corporate Responsibility
The Board takes regular account of the significance of social, environmental and ethical matters affecting the Group wherever it operates. It has developed a specific set of policies on corporate social responsibility, which seek to protect the interests of all of its stakeholders through ethical and transparent actions and include an anti-corruption policy and code of conduct.
Corporate Governance:
The Group is committed to high standards of corporate governance and seeks to continually evaluate its policies, procedures and structures to ensure that they are fit for purpose. In order to protect the interests of its shareholders and other stakeholders the Board has chosen to adopt the Quoted Companies Alliance (QCA) Corporate Governance Code for Small and mid-size Quoted Companies (the “QCA Code”), and the Directors are always
prepared, where practicable, to enter into dialogue with all such parties to promote a mutual understanding of objectives.
Full details of the Company's policy on Corporate Governance can be found on the website under:
https://probiotixhealth-ir.com/corporate/corporate-governance
Composition of the Board of Directors
The Board of Directors is currently comprised of the Chairman, Chief Executive Officer, Commercial Director, Executive Director and the three Non-Executive Directors.
Role of the Board:
The role of the Board is to agree the Group’s long-term strategy and direction and to monitor achievement of its business objectives. The Board meets several times per annum, either virtually or in person. Furthermore, it holds additional meetings as are necessary to transact ongoing business.
Board Committees:
Remuneration Committee
The Remuneration Committee is made up of Adam Reynolds, as Chairman with Marco Caspani has access to external expertise should that be required. This committee is responsible for the scale and structure of the remuneration of the Chief Executive, the Executive Directors and reports to the Chief Executive. The recommendations of the committee must be approved by the Board of Directors. No director or manager shall be involved in decisions relating to his/her own remuneration.
AQSE Rules Compliance Committee
The AQSE Rules Compliance Committee is chaired by Adam Reynolds. This committee is charged with ensuring that the Group has sufficient procedures, resources and controls in place to ensure compliance with the AQSE rules for companies. Among other things, the committee shall ensure that an Executive Director is at all times able to respond to requests for information from the Corporate Adviser and that all Directors and employees are aware of their obligations with regards to the disclosure of any trading in the Group’s shares.
Audit Committee
The Audit Committee, is chaired by Marco Caspani with Adam Reynolds. This committee is required to monitor the integrity of the financial statements of the Group, including the interim and annual reports. The committee also reviews financial returns to regulators and any financial information contained in announcements of a price sensitive nature. The committee shall also consider and make recommendations to the Board regarding resolutions to be put to shareholders for approval at the Annual General Meeting, with respect to the appointment or re-appointment of the Group’s external auditors. The Audit Committee, together with the external auditors, are responsible for determining the scope of the annual audit.
Nomination Committee
The Company does not currently have a nomination committee as the Board does not consider it appropriate to establish such a committee at this stage of the Company's development. Decisions which would usually be taken by the nomination committee will be taken by the Board as a whole.
Employees
The Group engages its employees in all aspects of the business and seeks to remunerate them fairly. The Group gives full and fair consideration to applications for employment regardless of age, gender, colour, ethnicity, disability, nationality, religious beliefs or sexual orientation. The Board takes employees’ interest into account when making decisions. Any suggestions from employees aimed at improving the Group’s performance are welcomed.
Suppliers and Contractors
The Group recognises that the goodwill of its contractors, consultants and suppliers is crucial to the success of its business and seeks to build and maintain this goodwill through fair and transparent business practices. The Group aims to settle genuine liabilities in accordance with contractual obligations.
Health and Safety
The Board recognises that it has a responsibility to provide strategic leadership and direction in the development and maintenance of the Group’s health and safety strategy, in order to protect all of its stakeholders
Section 172 Statement
In accordance with s172 of the Companies Act 2006 the Directors recognise their duty to promote the success of the company for the benefit of its stakeholders. Our commitment to delivering scientifically validated, safe, and effective probiotic-based microbiome solutions is underpinned by robust governance and a stakeholder-focused approach.
Stakeholder Engagement Our stakeholders include customers, employees, suppliers, investors, regulators, and the wider community. During the year, the Board actively engaged with each group through various initiatives:
- Customers - We continue to invest in scientific research and product innovation to uphold our promise of uncompromised safety and efficacy in microbiome health solutions. Market trends, customer feedback mechanisms and collaborations with leading institutions guide our development.
- Employees - The wellbeing and development of our employees remain central to our strategy. Initiatives such as involvement in strategic development, a flat management structure, freedom to plan securing a healthy work/life balance, training programs and employee engagement forums foster a positive workplace culture.
- Suppliers - Ethical sourcing and sustainability remain priorities. We work closely with supplier partnerships to ensure high-quality ingredients and sustainable supply chains, fostering long-term partnerships.
- Investors - Transparent communication and long-term value creation define our investor relations approach. Regular reporting and investor engagements demonstrate our strategic vision.
- Regulators - We adhere to the highest regulatory and compliance standards, ensuring product safety and scientific integrity. Strategic membership of industry leading organisations like the International Probiotic Association (IPA) ensures we remain aligned with current and future product regulation and legislation. Active engagement with regulatory bodies including AQUIS enables alignment with evolving industry standards and listing rules.
Long-Term Value and Sustainability
Recognising the importance of sustainability, we continue to integrate environmental and social responsibility into our business. Initiatives such as, responsible packaging, and ethical research practices support our long-term commitment to the health of people and the planet.
Board Decision-Making and Governance
The Board considers the interests of stakeholders in its decision-making processes. This includes evaluating risks, strategic opportunities, and industry developments to ensure resilient growth while maintaining ethical leadership.
We remain committed to upholding the principles of Section 172, ensuring that our actions contribute positively to society, safeguard stakeholder interests, and support sustainable long-term business success.
ON BEHALF OF THE BOARD
S Andersen
Date 27 March 2026
The Directors present their report and the audited financial statements of the group for the year to 31 December 2025.
PRINCIPAL ACTIVITY
The principal activity is that of developing probiotics to tackle cardiovascular metabolic health disease and other lifestyle conditions which are affecting growing numbers of people across the world.
DIRECTORS
The directors who served the company during the year and up to the date of this report were as follows:
Executive Directors
S Andersen
Non-executive Directors
M Caspani
A Reynolds
F Bruhn-Petersen
S P O’Hara
Directors’ Remuneration
Details of emoluments received by Directors of the Group for the year ended 31 December 2025 are as follows:
| Remuneration and fees |
Bonuses | Share based Payments |
Pension Costs |
Total | |
| £’000 | £’000 | £’000 | £’000 | £’000 | |
| A Reynolds* | 60,000 | - | 77,200 | - | 137,200 |
| S Andersen* | 264,218 | - | 198,246 | 12,938 | 475,402 |
| S P O’Hara* | 21,000 | - | 231,600 | - | 252,600 |
| M Caspani* | 21,000 | - | 7,000 | - | 28,000 |
| F Bruhn-Petersen* | 21,000 | - | - | - | 21,000 |
| Total | 387,218 | - | 514,046 | 12,938 | 914,202 |
Share based payments is an accounting charge and not remuneration paid to directors.
Directors and their interests
The directors of the group held the following beneficial interests in the shares and share options of Probiotix at the date of this report:
| Issued Share Capital | Share Options | |||
| Ordinary | Ordinary | Option | ||
| shares of | Percentage | shares of | exercise | |
| £0.0005 each | Held | £0.0005 each | Price (£) | |
| S P O’Hara | 6,131,450 | 3.88 | 3,000,000 | 0.210 |
| M Caspani | - | - | 500,000 | 0.210 |
| A Reynolds | 442,857 | 0.28 | 1,000,000 | 0.210 |
| S Andersen | 125,450 | 0.04 | 5,000,000 | 0.095 |
| F Bruhn-Petersen | 3,000,000 | 1.90 | - | - |
The share options held by S P O’Hara, and A Reynolds were granted on 31 March 2022 and are exercisable at £0.21 at any time up 30 March 2032, 50% of which vest on a doubling of the share price to £0.42 and 50% on an annual turnover of £2.5m
The share options held by M Caspani were granted on 31 March 2022 and are exercisable at £0.21 at any time up 30 March 2032, 1/3 of which vest on each of the 2-4 anniversaries of the date of grant.
The share options held by S Andersen were granted on 10 February 2025 and are exercisable at £0.095 at any time up 18 February 2035 50% vest when annual turnover reaches £2.5m, 50% vest on annual turnover of £5m.
Directors and Officers Insurance
The Group have a directors and Officers Insurance policy for £1m managed by CFC Underwriting Limited
SUBSTANTIAL SHAREHOLDINGS
Substantial shareholdings include directors as at 27 March 2026 were as follows:
| % of shares issued | |
| Optibiotix Health plc | 33.85 |
| Holdingselskabet of 29. Juni 2010 Aps. | 20.97 |
| Seneca Partners | 5.07 |
| Stephen O’Hara | 3.88 |
The share price per share at 31/12/2025 was £0.0775 (31/12/2024: £0.0625).
FINANCIAL INSTRUMENTS
The Group’s exposure to financial risk is set out in note 21 to the financial statements.
FINANCIAL AND CAPITAL RISK MANAGEMENT
The directors constantly monitor the financial risks and uncertainties facing the Group with particular reference to the exposure of credit risk and liquidity risk. They are confident that suitable policies are in place and that all material financial risks have been considered. The financial risk management objectives and policies can be found within note 21 of the financial statements.
The Board’s objective is to maintain a balance sheet that is both efficient and delivers long term shareholder value. The Group had cash balances of £1.27m at 31 December 2025 (2024: £1.65m) and had no short-term borrowings. The Board continues to monitor the balance sheet to ensure it has an adequate capital structure.
PRINCIPAL RISKS AND UNCERTAINTIES
| Market Risks | Impact | Mitigation |
| Economic uncertainty caused by changes in US policy | Ongoing uncertainty in trade relations following the change in the US administration | Manufacturing of some products has changed with effect from January 2026 to mitigate any major impacts on the business of the tariff regime. There was a small charge for tariff expenditure in 2025 £14k generally the business has been unaffected. |
| Economic uncertainty caused by war in Ukaine | Ongoing economic uncertainty, recession or an escalation of the war in Ukraine may impact market confidence, demand and prices. | The group is not directly affected by the war in Ukraine, but the Board monitor the general economic environment and consider economic forecasts when taking key decisions. The impact of the war in the middle east is under consideration and management will take appropriate action |
| Technology | The Group’s platform is currently unique. Rapid technological advances could see competitor products being launched. | The Group has product development plans in place for improved technology as well as for a wider product portfolio that includes additional innovative solutions for the targeted consumer groups. |
| Financial Risks | Impact | Mitigation |
| Future funding requirements | Potential as yet unidentified opportunities may not be pursued with the existing funding. | Management will analyse major opportunities and present them in additional business cases when warranted. The Company is able to sell its listed investments and raise further equity and debt finance. |
| Legal Risks | Impact | Mitigation |
| Intellectual Property litigation | Any claim brought against us would detract the Company from its business and incur potentially significant costs in defending its IP. | The Group engages with IP specialists to ensure we have a strong position. To our knowledge we do not infringe on any patents. |
| Compliance risks | The Group operates in many jurisdictions across the world. Local compliance issues may have an impact on the company. | Company maintains strong relationships with clients and monitors changes in local compliance laws which may have an impact. |
PRINCIPAL RISKS AND UNCERTAINTIES (Continued)
| Operational Risks | Impact | Mitigation |
| Loss of key personnel | Material adverse impact on the Group’s financial condition and prospects. | Competitive remuneration and option packages to reduce market volatility. The remuneration committee oversees the level of remuneration to ensure it remains competitive. |
| Technology | The Group is commercialising its technology to launch new products in the consumer market. | The Group has identified a need and responded to consumer demand. |
| Commercialisation | The Group has relied on sales to one customer in the early stages of commercialisation. A change in that customer outlook would affect Group sales growth. | The Group recruited experienced sales management team who have delivered a broad range of new customers to mitigate the reliance on one customer. |
| Cyber attacks | Cyber-attacks could delay or impair operations as which would have financial implications. | Training, anti-virus software, all users have multifactor authorisation for accounts, weekly review of attempts |
RESEARCH AND DEVELOPMENT
The Chairman and Chief Executive’s Statement gives information on the Group’s research and development activities.
EVENTS AFTER THE REPORTING PERIOD
There are no events after the reporting date.
PUBLICATION OF ACCOUNTS ON GROUP WEBSITE
Financial statements are published on the Group’s website. The maintenance and integrity of the website is the responsibility of the Directors. The Directors’ responsibilities also extend to the financial statements contained therein.
GOING CONCERN
The financial statements have been prepared on the assumption that the Group is a going concern. When assessing the foreseeable future, the Directors have looked at the Cash forecast for the next 12 months from the date of this report, the cash at bank available as at the date of approval of this report and are satisfied that the Group should be able to cover its committed cost, and other administrative expenses, as well as its ongoing research and development expenditure
After assessing the current loss making position, plausible downside scenarios, and based on strong revenue and forecasts, the directors have a reasonable expectation that the Group and the Company have adequate resources to continue in operational existence for the foreseeable future. Accordingly, they continue to adopt a going concern basis in preparing the annual report and financial statements.
STATEMENT OF DIRECTORS’ RESPONSIBILITIES
The Directors are responsible for preparing the annual report and the financial statements in accordance with applicable law and regulations. Company law requires the directors to prepare financial statements for each financial period. Under that law the directors have elected to prepare financial statements in accordance with UK adopted international accounting standards.
Under Company law the Directors must not approve the financial statements unless they are satisfied that they give a true and fair view of the state of affairs of the Group and Parent Company as at the end of the financial year and of the profit and loss of the Group for that period. In preparing these financial statements, the Directors are required to:
- select suitable accounting policies and then apply them consistently;
- make judgements and accounting estimates that are reasonable and prudent;
- state whether the Group and Parent Company financial statements have been prepared in accordance with UK adopted international accounting standards subject to any material departures disclosed and explained in the financial statements;
- prepare the financial statements on a going concern basis unless it is inappropriate to presume that the Group and Parent Company will continue in business.
The Directors confirm that they have complied with the above requirements in preparing the financial statements.
The Directors are responsible for keeping adequate accounting records that are sufficient to show and explain the Group’s and Parent Company’s transactions and disclose with reasonable accuracy at any time the financial position of the Group and Parent Company to enable them to ensure that the financial statements comply with the Companies Act 2006. They are also responsible for safeguarding the assets of the Company and Group and hence for taking reasonable steps for the prevention and detection of fraud and other irregularities.
The Directors are responsible for the maintenance and integrity of the corporate and financial information included on the Group’s website. Legislation in the United Kingdom governing the preparation and dissemination of financial statements and other information included in Directors’ Reports may differ from legislation in other jurisdictions.
STATEMENT AS TO DISCLOSURE OF INFORMATION TO AUDITORS So far as the Directors are aware, there is no relevant audit information (as defined by Section 418 of the Companies Act 2006) of which the Group and Parent Company auditor is unaware, and each Director has taken all the steps that he ought to have taken as a Director in order to make himself aware of any relevant audit information and to establish that the Group and Parent Company auditor is aware of the information.
AUDITOR
Gerald Edelman LLP has indicated its willingness to continue in office as auditor of the Company, and a resolution to re-appoint Gerald Edelman LLP will be proposed at the forthcoming Annual General Meeting.
STRATEGIC REPORT
In accordance with section 414C(11) of the Companies Act 2006 the Group chooses to report the future outlook and the risks and uncertainties faced by the Group in the Strategic Report.
ON BEHALF OF THE BOARD
S Andersen
Date 27 March 2026
Opinion
We have audited the financial statements of Probiotix Health PLC (the 'parent company') and its subsidiaries (the 'group') for the year ended 31 December 2025 which comprise the consolidated statement of comprehensive income, consolidated and company statements of financial position, consolidated and company statements of changes in equity, consolidated and company statement of cash flow, and notes to the consolidated and company financial statements, including a summary of significant accounting policies.
The financial reporting framework that has been applied in the preparation of the group financial statements is applicable law and UK adopted International Accounting Standards in conformity with the requirements of the Companies Act 2006. The financial reporting framework that has been applied in the preparation of the parent company financial statements is applicable law and United Kingdom Adopted International Accounting Standards.
In our opinion:
- the financial statements give a true and fair view of the state of the group's and of the parent company's affairs as at 31 December 2025 and of the group's loss for the year then ended;
- the group financial statements have been properly prepared in accordance with UK adopted International Accounting Standards;
- the parent company financial statements have been properly prepared in accordance with UK adopted International Accounting Standards in conformity with the requirements of the Companies Act 2006; and
- the financial statements have been prepared in accordance with the requirements of the Companies Act 2006.
Basis for opinion
We conducted our audit in accordance with International Standards on Auditing (UK) (ISAs (UK)) and applicable law. Our responsibilities under those standards are further described in the Auditor's responsibilities for the audit of the financial statements section of our report. We are independent of the Group and company in accordance with the ethical requirements that are relevant to our audit of the financial statements in the UK, including the FRC's Ethical Standard as applied to listed entities, and we have fulfilled our other ethical responsibilities in accordance with these requirements. We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our opinion.
Independence
We remain independent of the Group and the Company in accordance with the ethical requirements that are relevant to our audit of financial statements in the UK, including the FRC's Ethical Standard as applied to listed entities, and we have fulfilled our other ethical responsibilities in accordance with these requirements.
Conclusions relating to going concern
In auditing the financial statements, we have concluded that the director's use of the going concern basis of accounting in the preparation of the financial statements is appropriate. Our evaluation of the directors' assessment of the Group and parent company’s ability to continue to adopt the going concern basis of accounting included reviews of cash reserves and critical review of forecasts for a period of 12 months from when the financial statements are authorised for issue.
Based on the work we have performed, we have not identified any material uncertainties relating to events or conditions that, individually or collectively, may cast significant doubt on the group's ability to continue as a going concern for a period of at least twelve months from when the financial statements are authorised for issue.
Our responsibilities and the responsibilities of the directors with respect to going concern are described in the relevant sections of this report.
Overview
| Coverage | 100% of Group loss before tax 100% of Group total assets |
| Key audit matters | Revenue recognition has been identified as a key audit matter this year. This is essentially due to material increase in revenue through expansion in new geographies this year, and material revenue balance generated from a small number of high value customers. Impairment review of intangible assets (development cost and patents) and Going concern were key audit matters in the last year, however, in the current year these were not assessed and concluded as a key audit matter. This is based on our audit of impairment indicators review performed by management not resulting in any indicators requiring a full impairment review and our audit of going concern cash forecast prepared by management showing a strong net cash position. |
| Materiality | Group financial statements as a whole £41,000 (£33,000: 2024) based on 1.5% of gross assets. Company financial statements as a whole £20,000 (£14,000: 2024) based on 1.5% of gross assets. |
An overview of the scope of our audit
As part of designing our audit, we determined materiality and assessed the risks of material misstatement in the financial statements. In particular, we looked at where the directors made subjective judgments, for example in respect of significant accounting estimates that involved making assumptions and considering future events that are inherently uncertain. As in all of our audits we also addressed the risk of management override of internal controls, including evaluating whether there was evidence of bias by the directors that represented a risk of material misstatement due to fraud.
How we tailored the audit scope
We tailored the scope of our audit to ensure that we performed enough work to be able to give an opinion on the financial statements as a whole, taking into account the structure of the Group and the Company, the accounting processes and controls, and the industry in which they operate.
The Company and Probiotix Limited are significant components and were subject to full scope audit procedures by the Group audit team. Our audit scope for the non-significant components included performance of analytical review procedures. We also performed specified audit procedures over certain account balances and transaction classes that were regarded as material to the Group with a view to improve overall group audit coverage.
Key audit matters
Key audit matters are those matters that, in our professional judgement, were of most significance in our audit of the financial statements of the current period and include the most significant assessed risks of material misstatement (whether or not due to fraud) we identified, including those which had the greatest effect on, the overall audit strategy, the
allocation of resources in the audit; and directing the efforts of the engagement team. These matters were addressed in the context of our audit of the financial statements as a whole, and in forming our opinion thereon, and we do not provide a separate opinion on these matters.
| Key audit matter | How our audit addressed the key audit matter |
| Revenue recognition The group has recognized £2.7 million of revenue this year which has been identified as a key audit matter. This is essentially due to material increase in revenue through expansion in new geographies this year, and material revenue balance generated from a small number of high value customers. The primary revenue stream arises from the sale of products to business customers. There is a risk as to whether the revenue recognised meets the revenue recognition criteria under IFRS 15, particularly on the occurrence of the transaction or if an appropriate cut-off is applied in this regard. There is also a risk if the Group’s accounting policy is compliant with the requirements of IFRS. Refer to Accounting Policies, Note 2.1 |
We have performed the following audit procedures:
Key observations: |
Our application of materiality
Materiality is assessed as the magnitude of an omission or misstatement that, individually or in the aggregate, could reasonably be expected to influence the economic decisions of the users of the financial statements. Misstatements below these levels will not necessarily be evaluated as immaterial as we also take account of the nature of identified misstatements, and the particular circumstances of their occurrence, when evaluating their effect on the financial statements as a whole. Materiality provides a basis for determining the nature and extent of our audit procedures.
Based on our professional judgment, we determined materiality for the financial statements as a whole as follows:
| Overall materiality | £41,000 (£33,000: 2024) | £20,000 (£14,000: 2024) |
| How we determined it | 1.5% of revenue | 1.5 % of administrative expenses |
| Rationale for benchmark applied | We believe that gross assets is a primary measure used by shareholders in assessing the performance of the group. | We believe that gross assets is a primary measure used by shareholders in performance of the Company as the holding company within the Group. |
| Performance materiality | £26,000 (£21,000: 2024) | £13,000 (£9,000: 2024) |
| Basis for determining performance materiality | 65% of materiality. In reaching our conclusion on the level of performance materiality to be applied we considered a number of factors including the expected total value of known and likely misstatements (based on past experience), our knowledge of the Group’s control environment and management’s attitude towards proposed adjustments. | 65% of materiality. In reaching our conclusion on the level of performance materiality to be applied we considered a number of factors including the expected total value of known and likely misstatements (based on past experience), our knowledge of the Group’s control environment and management’s attitude towards proposed adjustments. |
Component materiality
For each component in the scope of our Group audit, we allocated a materiality that is equal to or less than our overall Group materiality. The range of materiality allocated across components ranged from £16,400 to £36,900. We set materiality for each significant component of the Group based on a percentage of between 40% and 92% of Group materiality dependent on the size and our assessment of the risk of material misstatement of that component. In the audit of each component, we further applied performance materiality levels of 65% of the component materiality to our testing to ensure that the risk of errors exceeding component materiality was appropriately mitigated.
Reporting threshold
We agreed with the Audit Committee that we would report to them misstatements identified during our audit above £1,600 for the both the Group and £1,000 Company audit as well as misstatements below those amounts that, in our view, warranted reporting for qualitative reasons.
Other information
The directors are responsible for the other information contained within the annual report. The other information comprises the information included in the annual report, other than the financial statements and our auditor's report thereon. Our opinion on the financial statements does not cover the other information and, except to the extent otherwise explicitly stated in our report, we do not express any form of assurance conclusion thereon.
In connection with our audit of the financial statements, our responsibility is to read the other information and, in doing so, consider whether the other information is materially inconsistent with the financial statements or our knowledge obtained in the audit or otherwise appears to be materially misstated. If we identify such material inconsistencies or apparent material misstatements, we are required to determine whether there is a material misstatement in the financial statements or a material misstatement of the other information. If, based on the work we have performed, we conclude that there is a material misstatement of this other information, we are required to report that fact.
We have nothing to report in this regard.
Opinions on other matters prescribed by the Companies Act 2006
In our opinion, based on the work undertaken in the course of the audit:
- the information given in the strategic report and the directors' report for the financial year for which the financial statements are prepared is consistent with the financial statements; and
- the strategic report and the directors' report have been prepared in accordance with applicable legal requirements.
Matters on which we are required to report by exception
In the light of the knowledge and understanding of the group and Company and its environment obtained in the course of the audit, we have not identified material misstatements in the strategic report or the directors' report.
We have nothing to report in respect of the following matters in relation to which the Companies Act 2006 requires us to report to you if, in our opinion:
- adequate accounting records have not been kept by the Group and Company, or returns adequate for our audit have not been received from branches not visited by us; or
- the Group and Company financial statements are not in agreement with the accounting records and returns; or
- certain disclosures of directors' remuneration specified by law are not made; or
- we have not received all the information and explanations we require for our audit.
Responsibilities of directors
As explained more fully in the Statement of Directors' responsibilities statement set out on page 19, the directors are responsible for the preparation of the financial statements and for being satisfied that they give a true and fair view, and for such internal control as the directors determine is necessary to enable the preparation of financial statements that are free from material misstatement, whether due to fraud or error.
In preparing the financial statements, the directors are responsible for assessing the Group's and Company's ability to continue as a going concern, disclosing, as applicable, matters related to going concern and using the going concern basis of accounting unless the directors either intend to liquidate the Group or the Company or to cease operations, or have no realistic alternative but to do so.
Auditor's responsibilities for the audit of the financial statements
The objectives of our audit, in respect to fraud are; to identify and assess the risks of material misstatement of the financial statements due to fraud; to obtain sufficient appropriate audit evidence regarding the assessed risks of material misstatements due to fraud, through designing and implementing appropriate responses; and to respond appropriately to fraud or suspected fraud identified during the audit. However, the primary responsibility for the prevention and detection of fraud rests with both those charged with governance of the entity and management.
Our audit procedures were primarily directed towards testing the accounting systems in operation upon which we have based our assessment of the financial statements for the year-ended 31 December 2025.
Our approach to identifying and assessing the risks of material misstatement in respect of irregularities, including fraud and non-compliance with laws and regulations, was as follows:
- the engagement partner ensured the engagement team collectively had the appropriate competence, capabilities and skills to identify or recognise non-compliance with applicable laws and regulations in the United Kingdom;
- we identified the laws and regulations applicable to the company through discussions with directors and other management, and from our knowledge and experience of the entity's activities.
- we focused on specific laws and regulations which we considered may have a direct material effect on the financial statements or the operations of the company, including Companies Act 2006, taxation legislation, data protection, employment and health and safety legislation.
- we assessed the extent of compliance with the laws and regulations identified above through making enquiries of management and reviewing legal expenditure; and
- identified laws and regulations were communicated within the audit team regularly and the team remained alert to instances of non-compliance throughout the audit.
We assessed the susceptibility of the company's financial statements to material misstatement, including obtaining an understanding of how fraud might occur, by:
- making enquiries of management as to where they considered there was susceptibility to fraud, their knowledge of actual, suspected and alleged fraud; and
- considering the internal controls in place to mitigate risks of fraud and non-compliance with laws and regulations.
To address the risk of fraud through management bias and override of controls, we:
- performed analytical procedures to identify any unusual or unexpected relationships;
- tested journal entries to identify unusual transactions;
- assessed whether judgements and assumptions made in determining the accounting estimates as disclosed in Note 2 of the financial statements were indicative of potential bias; and
- investigated the rationale behind significant or unusual transactions.
In response to the risk of irregularities and non-compliance with laws and regulations, we designed procedures which included, but were not limited to:
- reading the minutes of meetings of those charged with governance; and
- enquiring of management as to actual and potential litigation and claims.
There are inherent limitations in our audit procedures described above. The more removed that laws and regulations are from financial transactions, the less likely it is that we would become aware of non-compliance.
Auditing standards also limit the audit procedures required to identify noncompliance with laws and regulations to enquiry of the directors and other management and the inspection of regulatory and legal correspondence, if any.
Material misstatements that arise due to fraud can be harder to detect than those that arise from error as they may involve deliberate concealment or collusion.
A further description of our responsibilities for the audit of the financial statements is located on the Financial Reporting Council's website at: http://www.frc.org.uk/auditorsresponsibilities
This description forms part of our auditor's report.
Use of this report
This report, including the opinions, has been prepared for and only for the Company's members as a body in accordance with Chapter 3 of Part 16 of the Companies Act 2006 and for no other purpose. We do not, in giving these opinions, accept or assume responsibility for any other purpose or to any other person to whom this report is shown or into whose hands it may come save where expressly agreed by our prior consent in writing.
Talha Farrukh
Audit Partner
For and on behalf of Gerald Edelman LLP,
Senior Statutory Auditor
73 Cornhill
London United Kingdom
EC3V 3QQ
27 March 2026
Consolidated Statement of Comprehensive Income
For the year ended 31 December 2025
| Notes | Year ended 31 December 2025 |
Year ended 31 December 2024 |
|||
| £’000 | £’000 | ||||
| Revenue from contracts with customers | 3 | 2,732 | 1,883 | ||
| Cost of sales | (1,250) | (886) | |||
| ─────── | ─────── | ||||
| Gross Profit | 1,482 | 997 | |||
| Share based payment cost | (753) | (26) | |||
| Depreciation and amortisation | (54) | (68) | |||
| Other administrative costs | (1,910) | (1,755) | |||
| Total administrative expenses | 5 | (2,717) | (1,849) | ||
| ─────── | ─────── | ||||
| Operating loss | (1,235) | (852) | |||
| ─────── | ─────── | ||||
| Loss before tax | (1,235) | (852) | |||
| Taxation | 6 | 2 | 5 | ||
| ─────── | ─────── | ||||
| Loss for the period | (1,233) | (847) | |||
| Other Comprehensive Income | - | - | |||
| ─────── | ─────── | ||||
| Total comprehensive loss for the period | |
(1,233) |
(847) |
||
| ═══════ | ═══════ | ||||
| Total comprehensive loss attributable to: Owners of the company | (1,233) | (847) | |||
| ═══════ | ═══════ | ||||
| Earnings per share from continued operations | |||||
| Basic profit/(loss) per share - pence | 7 | (0.78)p | (0.63)p | ||
| Diluted profit/(loss) per share - pence | (0.71)p | (0.61)p | |||
| ═══════ | ═══════ |
All activities relate to continuing operations
Consolidated Statement of Financial Position
As at 31 December 2025
| Notes | As at 31 December 2025 |
As at 31 December 2024 |
|||
| ASSETS | £’000 | £’000 | |||
| Non-current assets | |||||
| Intangibles | 8 | 186 | 236 | ||
| Property, plant and equipment | 9 | 5 | 7 | ||
| ─────── | ─────── | ||||
| 191 | 243 | ||||
| ─────── | ─────── | ||||
| CURRENT ASSETS | |||||
| Inventories | 11 | - | 31 | ||
| Trade and other receivables | 12 | 351 | 257 | ||
| Cash and cash equivalents | 13 | 1,266 | 1,646 | ||
| ─────── | ─────── | ||||
| 1,617 | 1,934 | ||||
| ─────── | ─────── | ||||
| TOTAL ASSETS | 1,808 | 2,177 | |||
| ═══════ | ═══════ | ||||
| EQUITY | |||||
| Shareholders’ Equity | |||||
| Called up share capital | 14 | 79 | 79 | ||
| Share premium | 15 | 4,534 | 4,534 | ||
| Share based payment reserve | 15 | 794 | 41 | ||
| Group reorganisation reserve | 15 | (945) | (945) | ||
| Retained earnings | 15 | (3,019) | (1,786) | ||
| ─────── | ─────── | ||||
| Total Equity | 1,443 | 1,923 | |||
| ─────── | ─────── | ||||
| LIABILITIES | |||||
| Current liabilities | |||||
| Trade and other payables | 16 | 318 | 194 | ||
| ─────── | ─────── | ||||
| 318 | 194 | ||||
| ─────── | ─────── | ||||
| Non - current liabilities | |||||
| Deferred tax liability | 17 | 47 | 60 | ||
| ─────── | ─────── | ||||
| 47 | 60 | ||||
| ─────── | ─────── | ||||
| TOTAL LIABILITIES | 365 | 254 | |||
| ─────── | ─────── | ||||
| TOTAL EQUITY AND LIABILITIES | 1,808 | 2,177 | |||
| ═══════ | ═══════ |
These financial statements were approved and authorised for issue by the Board of Directors on 30 March 2026 and were signed on its behalf by:
S Andersen
Director
Company Registration no. 13723211
Consolidated Statement of Changes in Equity
For the period ended 31 December 2025
|
Called up Share capital |
Share Premium |
Share-based Payment Reserve |
Group Reorganisation Reserve |
Retained Earnings |
Total equity |
|
| £’000 | £’000 | £,000 | £’000 | £’000 | £’000 | |
| As at 1 January 2024 | 61 | 3,338 | 57 | (945) | (980) | 1,531 |
| Loss for the period | - | - | - | - | (847) | (847) |
| Forfeiture of share options | - | - | (41) | - | 41 | - |
| Share based payments | - | - | 25 | - | - | 25 |
| Share issue | 18 | 1,208 | - | - | - | 1,226 |
| Share issue costs | (12) | (12) | ||||
| ────── | ─────── | ────── | ────── | ───── | ─────── | |
| Balance at 31 December 2024 | 79 | 4,534 | 41 | (945) | (1,786) | 1,923 |
| Loss for the period | - | - | - | - | (1,233) | (1,233) |
| Forfeiture of share options | - | - | - | - | - | |
| Share based payment | - | - | 753 | - | - | 753 |
| Share issue | - | - | - | - | - | - |
| Share issue costs | - | - | ||||
| ────── | ─────── | ────── | ────── | ───── | ─────── | |
| Balance at 31 December 2025 | 79 | 4,534 | 794 | (945) | (3,019) | 1,443 |
| ══════ | ═══════ | ══════ | ══════ | ═════ | ═══════ |
The notes form part of these financial statements
Consolidated Statement of Cash Flows
For the period ended 31 December 2025
| Notes | Year ended 31 December 2025 |
Year ended 31 December 2024 |
||
| £ | £ | |||
| Cash flows from operating activities | ||||
| Cash utilised by operations | 1 | (367) | (1,060) | |
| ────── | ────── | |||
| Net cash outflow from operating activities | (367) | (1,060) | ||
| Cash flows from investing activities | ||||
| Acquisition of property, plant and equipment | (12) | (10) | ||
| ────── | ────── | |||
| Net cash outflow from investing activities | (12) | (10) | ||
| ────── | ────── | |||
| Cash flows from financing activities | ||||
| Share issues net of issue costs | - | 1,214 | ||
| ────── | ────── | |||
| Net cash inflow from financing activities | - | 1,214 | ||
| ────── | ────── | |||
| Increase/(decrease) in cash and equivalents | (380) | 144 | ||
| Cash and cash equivalents at beginning of period | 1,646 | 1,502 | ||
| ────── | ────── | |||
| Cash and cash equivalents at end of period | 2 | 1,266 | 1,646 | |
| ══════ | ══════ | |||
